The hypothalamic–pituitary–adrenal (HPA) axis governs our central stress response system. It reflects the dynamic intertwining of the central nervous system (CNS) and endocrine system and defines the tight hormonal coupling of the hypothalamus, the anterior pituitary and the adrenal cortex. The HPA axis is controlled by a set of direct influences and feedback interactions that governs the adaptation response to stress and regulates several critical physical processes encompassing the digestive system, the immune system, sexuality, mood and energy balance.
When the HPA axis is activated in response to stress it impacts the balance of the neurotransmitters dopamine, serotonin, norepinephrine, gamma-aminobutyric acid (or GABA) and glutamate, and also modulates and impacts insulin sensitivity and the balance of T3:T4 (or triiodothyronine and thyroxine) thyroid hormones.

Highlights
- HPA axis activates in response to stress it impacts the balance of the neurotransmitters dopamine, serotonin, norepinephrine, gamma-aminobutyric acid (or GABA) and glutamate,
- Cortisol regulates the immune system, the digestive system and the reproductive system. During non-stressful situations, a certain level of cortisol is maintained in the bloodstream.
The stress response, as facilitated by the HPA axis, is characterized by neurons in the paraventricular nucleus (PVN) of the hypothalamus releasing two neurohormones—corticotropin-releasing factor (CRF) and arginine vasopressin (AVP)—into the blood vessels connecting the hypothalamus and the pituitary gland (i.e. hypophysial portal blood). CRF and AVP stimulate the anterior pituitary gland to produce and secrete adrenocorticotropic hormone (ACTH) into the bloodstream. The ACTH then induces synthesis of glucocorticoids (steroid hormones) and releases it from the adrenal glands, which are located atop the kidneys. The main glucocorticoid in humans is cortisol.
The hypothalamic activation of the HPA axis is modulated by various brain signalling (i.e. neurotransmitter) systems. As stated in a study, “[s]ome of these systems have inhibitory effects (e.g., γ-aminobutyric acid [GABA] and opioids), whereas others have excitatory effects (e.g., norepinephrine and serotonin) on the PVN. Thus, the CNS and the hormone (i.e. endocrine) systems are tightly interconnected to coordinate glucocorticoid activity.
Cortisol, stress and the HPA axis
The main role of cortisol is to release glucose into the bloodstream in order to facilitate the “flight or fight” response. It also regulates the immune system, the digestive system and the reproductive system. During non-stressful situations, a certain level of cortisol is maintained in the bloodstream. The circadian rhythm of ACTH and cortisol release reaches the highest levels around 8–10 a.m. in the morning, while the lowest levels are recorded around midnight.’
Stress results in an increase in overall cortisol output. When there is a chronic stressful situation, changes occur to the neurons in the PVN and other areas in the brain, which results in increased sustained activation of the HPA axis. Long-term stress can result in negative feedback to the HPA axis, resulting in burnout. Chronic stress can result in high or low cortisol output—this depends on the nature of the threat, the time elapsed since the onset of said threat as well as the person’s response to the situation.
HPA axis dysfunction
HPA axis dysfunction is caused due to continuous or sudden stress and a variation of the stress response, resulting in a dysregulation of stress hormones—mainly an alteration in the quantity and/or diurnal pattern of adrenal hormone secretion, including cortisol and dehydroepiandrosterone (DHEA, a steroid hormone produced in the adrenal glands).
Data reveals that more than 50 percent of Americans suffer from one or more chronic conditions, including major depressive disorder, generalized anxiety disorder, low testosterone, estrogen imbalances, diabetes , hypertension, autoimmune disorders, chronic pain, metabolic syndrome, cardiovascular disease and hypothyroid, all of which are associated with disturbances of the HPA axis.
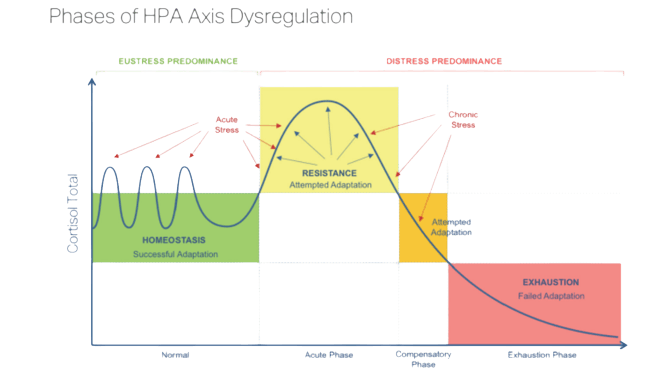
HPA axis dysfunction may begin all of a sudden, or as a result of periods of prolonged, repeated stress. The source of this stress may be positive or negative, and it may or may not be acknowledged by the individual. It can also be caused by recurrent disease and illness, physical stress like injury, changes in diet, surgery, tobacco/alcohol addiction, etc emotional stressors like grief or loss, and environmental stressors like the chemical pollution of air, water, food, etc.
Conclusion
In the case of athletes, HPA axis dysfunction leads to a decrease in sport-specific performance associated with fatigue, termed ‘overtraining syndrome’. Inadequate recovery, low calorie intake resulting in negative energy balance, social pressures and excessive progression in training volume and intensity are the key triggers for this. Studies indicate that impaired hormonal responses due to chronic exposure to stress could decrease the responsiveness of the HPA axis in athletes.
The adverse effects of HPA axis dysfunction can be rectified through lifestyle modifications including exercise, inculcating healthy sleep patterns, balanced diet, stress management, avoidance of caffeine, alcohol, refined sugars and being vigilant about any food allergies/sensitivities. Individualised treatment plans may include supplementation of dietary cofactors necessary for mitigating the stress response and cortisol production.
Disclaimer: The contents of this article are for general information and educational purposes only. It neither provides any medical advice nor intends to substitute professional medical opinion on the treatment, diagnosis, prevention or alleviation of any disease, disorder or disability. Always consult with your doctor or qualified healthcare professional about your health condition and/or concerns and before undertaking a new healthcare regimen including making any dietary or lifestyle changes.
References